Figure 2626b shows carbon (C) K edge from materials containing the carbonate anion (calcite, siderite, desautelsite, and hydrotalcite) and three allotropes of C (amorphous, graphite, and diamond). In the valence band, valence electron transitions into the π* states also produce a peak at around 6 eV. For graphite, the transitions of the inner shell electrons into unoccupied π* states give a peak prior to the edge onset. For instance, in core loss spectra from diamond, the excitation of the 1s electrons to the σ* states generates the carbon k edge peak. 
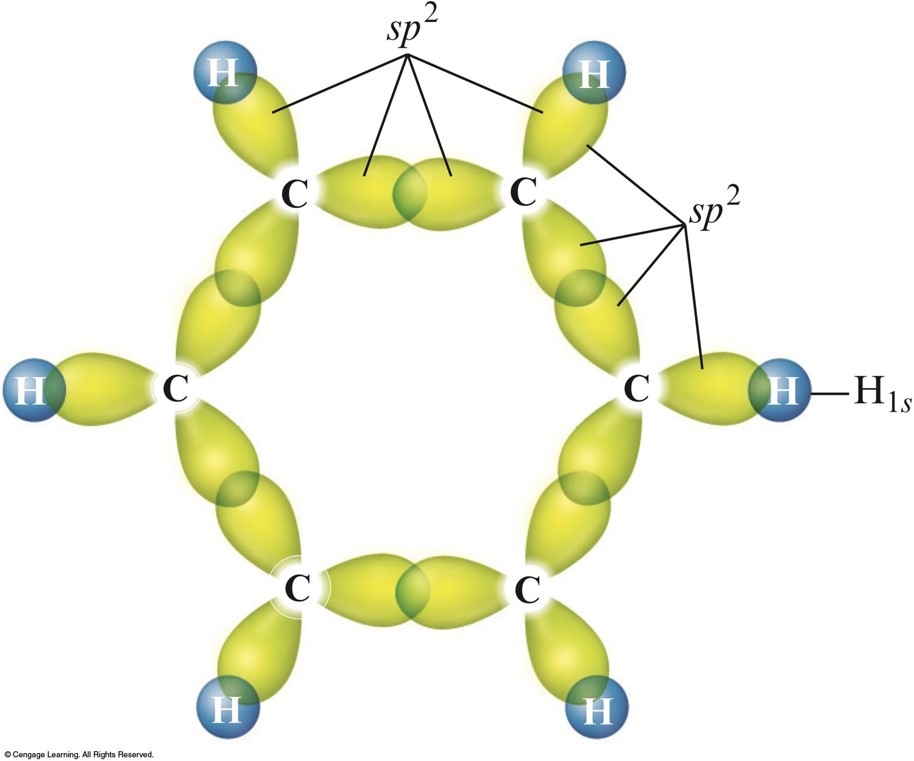
For the diamond and graphite, electron transitions to these states generate many of the characteristic features in EEL spectra. The sp 2 bonded solids have both σ/σ* and π/π* states available to the electrons, while for sp 3 bonded solids only the σ/ σ* states present. Each carbon atom in diamond forms a series of sp 3 hybridised atomic orbitals. Schematic illustration of the process of hybridisation for carbon. In this case, these four valence electrons form σ bonds.įigure 2626a. During this process, the energy levels of the 2s are raised while the 2p levels are reduced, resulting in a more favourable lower energy system. Figure 2626a illustrates the hybridization process for diamond. Graphite is one example with an sp 2 type, while diamond is of an sp 3 form. 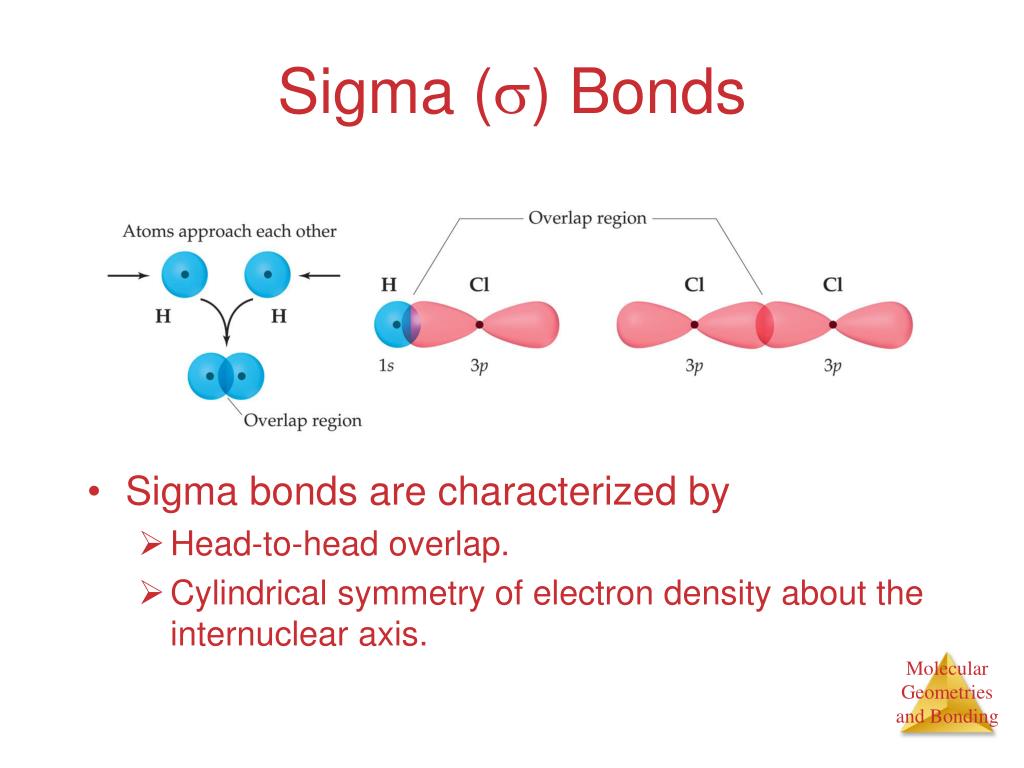
The configuration of the valence electrons in carbon is a combination (called hybrids) of s and p orbitals. In chemistry, sigma bonds (σ bonds) are actually the strongest type of covalent chemical bonds. The sigma (σ) bonds are formed by axial overlap of half-filled atomic orbitals of atoms taking part in bonding. This book (Practical Electron Microscopy and Database) is a reference for TEM and SEM students, operators, engineers, technicians, managers, and researchers.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
